What Specifically Happens to a Cell During the Process of Apoptosis
The cells of a multicellular organism are members of a highly organized community. The number of cells in this community is tightly regulated—not simply by controlling the rate of cell division, merely also by controlling the rate of cell expiry. If cells are no longer needed, they commit suicide past activating an intracellular death program. This process is therefore called programmed prison cell death, although it is more commonly called apoptosis (from a Greek word meaning "falling off," every bit leaves from a tree).
The corporeality of apoptosis that occurs in developing and adult creature tissues tin be astonishing. In the developing vertebrate nervous organisation, for example, upwards to half or more of the nerve cells normally die before long after they are formed. In a healthy developed human, billions of cells die in the bone marrow and intestine every hour. It seems remarkably wasteful for and then many cells to die, specially as the vast majority are perfectly healthy at the time they kill themselves. What purposes does this massive jail cell death serve?
In some cases, the answers are articulate. Mouse paws, for example, are sculpted by jail cell decease during embryonic development: they start out every bit spadelike structures, and the private digits dissever only equally the cells between them dice (Effigy 17-35). In other cases, cells die when the structure they form is no longer needed. When a tadpole changes into a frog, the cells in the tail dice, and the tail, which is not needed in the frog, disappears (Effigy 17-36). In many other cases, prison cell expiry helps regulate cell numbers. In the developing nervous system, for case, cell expiry adjusts the number of nerve cells to friction match the number of target cells that require innervation. In all these cases, the cells dice by apoptosis.

Figure 17-35
Sculpting the digits in the developing mouse paw by apoptosis. (A) The paw in this mouse embryo has been stained with a dye that specifically labels cells that have undergone apoptosis. The apoptotic cells appear equally brilliant green dots betwixt the developing (more...)

Figure 17-36
Apoptosis during the metamorphosis of a tadpole into a frog. Every bit a tadpole changes into a frog, the cells in the tadpole tail are induced to undergo apoptosis; every bit a result, the tail is lost. All the changes that occur during metamorphosis, including (more...)
In adult tissues, cell decease exactly balances cell sectionalization. If this were not so, the tissue would grow or compress. If part of the liver is removed in an adult rat, for example, liver jail cell proliferation increases to make upwards the loss. Conversely, if a rat is treated with the drug phenobarbital—which stimulates liver cell segmentation (and thereby liver enlargement)—and and so the phenobarbital treatment is stopped, apoptosis in the liver greatly increases until the liver has returned to its original size, usually within a week or so. Thus, the liver is kept at a abiding size through the regulation of both the jail cell death charge per unit and the jail cell birth rate.
In this brusk section, we describe the molecular mechanisms of apoptosis and its control. In the final section, nosotros consider how the extracellular control of cell proliferation and prison cell death contributes to the regulation of cell numbers in multicellular organisms.
Apoptosis Is Mediated past an Intracellular Proteolytic Cascade
Cells that die as a upshot of acute injury typically swell and flare-up. They spill their contents all over their neighbors—a process called prison cell necrosis—causing a potentially dissentious inflammatory response. By dissimilarity, a prison cell that undergoes apoptosis dies neatly, without damaging its neighbors. The jail cell shrinks and condenses. The cytoskeleton collapses, the nuclear envelope disassembles, and the nuclear DNA breaks up into fragments. Most chiefly, the prison cell surface is altered, displaying properties that cause the dying cell to be rapidly phagocytosed, either by a neighboring cell or past a macrophage (a specialized phagocytic cell, discussed in Affiliate 24), earlier any leakage of its contents occurs (Effigy 17-37). This not only avoids the damaging consequences of cell necrosis but also allows the organic components of the expressionless cell to exist recycled by the cell that ingests it.
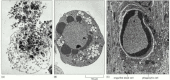
Figure 17-37
Cell death. These electron micrographs show cells that take died by (A) necrosis or (B and C) apoptosis. The cells in (A) and (B) died in a civilization dish, whereas the cell in (C) died in a developing tissue and has been engulfed by a neighboring prison cell. (more...)
The intracellular machinery responsible for apoptosis seems to be like in all animal cells. This mechanism depends on a family unit of proteases that have a cysteine at their active site and cleave their target proteins at specific aspartic acids. They are therefore called caspases. Caspases are synthesized in the jail cell equally inactive precursors, or procaspases, which are usually activated by cleavage at aspartic acids by other caspases (Figure 17-38A). One time activated, caspases cleave, and thereby activate, other procaspases, resulting in an amplifying proteolytic cascade (Figure 17-38B). Some of the activated caspases then cleave other central proteins in the cell. Some cleave the nuclear lamins, for example, causing the irreversible breakdown of the nuclear lamina; some other cleaves a protein that usually holds a Dna-degrading enzyme (a DNAse) in an inactive form, freeing the DNAse to cut up the Dna in the jail cell nucleus. In this fashion, the cell dismantles itself rapidly and neatly, and its corpse is rapidly taken up and digested by another jail cell.
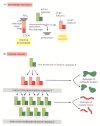
Effigy 17-38
The caspase cascade involved in apoptosis. (A) Each suicide protease is fabricated as an inactive proenzyme (procaspase), which is ordinarily activated by proteolytic cleavage by another member of the caspase family. As indicated, 2 of the cleaved fragments (more than...)
Activation of the intracellular cell death pathway, like entry into a new phase of the cell bike, is usually triggered in a complete, all-or-none fashion. The protease cascade is not but destructive and self-amplifying but also irreversible, then that once a prison cell reaches a critical point forth the path to destruction, information technology cannot plow back.
Procaspases Are Activated by Binding to Adaptor Proteins
All nucleated brute cells contain the seeds of their own destruction, in the course of diverse inactive procaspases that lie waiting for a bespeak to destroy the cell. It is therefore not surprising that caspase action is tightly regulated inside the jail cell to ensure that the death program is held in cheque until needed.
How are procaspases activated to initiate the caspase cascade? A general principle is that the activation is triggered past adaptor proteins that bring multiple copies of specific procaspases, known every bit initiator procaspases, close together in a circuitous or aggregate. In some cases, the initiator procaspases accept a small amount of protease activity, and forcing them together into a complex causes them to cleave each other, triggering their mutual activation. In other cases, the assemblage is idea to crusade a conformational change that activates the procaspase. Inside moments, the activated caspase at the pinnacle of the cascade cleaves downstream procaspases to amplify the death indicate and spread information technology throughout the cell (see Effigy 17-38B).
Procaspase activation can be triggered from outside the cell by the activation of death receptors on the prison cell surface. Killer lymphocytes (discussed in Affiliate 24), for instance, tin can induce apoptosis by producing a poly peptide chosen Fas ligand, which binds to the death receptor poly peptide Fas on the surface of the target cell. The clustered Fas proteins then recruit intracellular adaptor proteins that demark and aggregate procaspase-8 molecules, which cleave and activate i some other. The activated caspase-eight molecules and so actuate downstream procaspases to induce apoptosis (Figure 17-39A). Some stressed or damaged cells impale themselves past producing both the Fas ligand and the Fas protein, thereby triggering an intracellular caspase cascade.
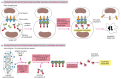
Figure 17-39
Induction of apoptosis by either extracellular or intracellular stimuli. (A) Extracellular activation. A killer lymphocyte carrying the Fas ligand binds and activates Fas proteins on the surface of the target cell. Adaptor proteins bind to the intracellular (more...)
When cells are damaged or stressed, they can also impale themselves past triggering procaspase aggregation and activation from within the cell. In the best understood pathway, mitochondria are induced to release the electron carrier protein cytochrome c (see Figure 14-26) into the cytosol, where it binds and activates an adaptor protein called Apaf-1 (Effigy 17-39B). This mitochondrial pathway of procaspase activation is recruited in most forms of apoptosis to initiate or to advance and amplify the caspase cascade. DNA harm, for instance, equally discussed earlier, can trigger apoptosis. This response usually requires p53, which can activate the transcription of genes that encode proteins that promote the release of cytochrome c from mitochondria. These proteins belong to the Bcl-ii family unit.
Bcl-ii Family unit Proteins and IAP Proteins Are the Master Intracellular Regulators of the Cell Death Programme
The Bcl-2 family of intracellular proteins helps regulate the activation of procaspases. Some members of this family, like Bcl-2 itself or Bcl-Ten L , inhibit apoptosis, at least partly past blocking the release of cytochrome c from mitochondria. Other members of the Bcl-2 family unit are not death inhibitors, but instead promote procaspase activation and jail cell expiry. Some of these apoptosis promoters, such every bit Bad, role by bounden to and inactivating the death-inhibiting members of the family, whereas others, similar Bax and Bak, stimulate the release of cytochrome c from mitochondria. If the genes encoding Bax and Bak are both inactivated, cells are remarkably resistant to virtually apoptosis-inducing stimuli, indicating the crucial importance of these proteins in apoptosis induction. Bax and Bak are themselves activated by other apoptosis-promoting members of the Bcl-2 family such as Bid.
Some other of import family of intracellular apoptosis regulators is the IAP (inhibitor of apoptosis) family. These proteins are thought to inhibit apoptosis in ii ways: they bind to some procaspases to forbid their activation, and they bind to caspases to inhibit their activity. IAP proteins were originally discovered every bit proteins produced by sure insect viruses, which use them to forestall the infected prison cell from killing itself earlier the virus has had fourth dimension to replicate. When mitochondria release cytochrome c to activate Apaf-1, they besides release a protein that blocks IAPs, thereby greatly increasing the efficiency of the death activation procedure.
The intracellular cell death program is too regulated by extracellular signals, which can either activate apoptosis or inhibit it. These signal molecules mainly act by regulating the levels or activity of members of the Bcl-two and IAP families. We encounter in the next section how these signal molecules aid multicellular organisms regulate their cell numbers.
Summary
In multicellular organisms, cells that are no longer needed or are a threat to the organism are destroyed by a tightly regulated cell suicide process known every bit programmed jail cell death, or apoptosis. Apoptosis is mediated by proteolytic enzymes called caspases, which trigger prison cell death by cleaving specific proteins in the cytoplasm and nucleus. Caspases exist in all cells as inactive precursors, or procaspases, which are usually activated by cleavage by other caspases, producing a proteolytic caspase cascade. The activation process is initiated by either extracellular or intracellular death signals, which cause intracellular adaptor molecules to amass and activate procaspases. Caspase activation is regulated past members of the Bcl-2 and IAP poly peptide families.

Source: https://www.ncbi.nlm.nih.gov/books/NBK26873/
0 Response to "What Specifically Happens to a Cell During the Process of Apoptosis"
Post a Comment